
A global economy needs to be certain that analytical data, generated in one country by one analytical laboratory, will be recognised and universally accepted. To achieve this aim international standards for laboratory quality systems have been introduced and subsequently developed. Together with a myriad of formal international mutual recognition agreements between national certification bodies, underpinned by ILAC and EA, the principle of mutual recognition of certified reference materials (CRMs) used to calibrate and control analytical systems is now well established.

With modern analytical chemistry required to deliver data that is accepted and understood on this global scale it is important that such data must show clear traceability to the internationally recognised SI units. Therefore in analytical chemistry, which deals with amounts of chemical substances reacting together, the SI unit to work with is clearly the mole, one of the seven fundamental units of measure that are defined by the SI System (SI = Système Internationale).
But what, exactly, is the mole? It was originally defined as the number of discrete particles (in this case atoms) contained within 12 grams of the pure isotope carbon-12. This is all very well and good, but the choice of carbon does create problems for analytical chemists because carbon is so unreactive. One of its forms, diamond, is the hardest known natural substance.
To get around the non-reactivity of carbon the only way to produce certified reference materials (CRMs) for trace element analysis has been to look at each element in isolation and thus produce separate elemental CRMs, each an individual realisation of the mole. This is an extremely costly and complex process, traditionally undertaken only by government funded national metrology institutes (NMIs). As a result, and even today, most trace element analyses are calibrated through reference materials provided by such NMIs who themselves do not have accreditation to the laboratory quality systems that other members of the analytical community are now expected to achieve as a matter of course.
During the late 1990s we at ROMIL identified a real need for a more practical and affordable scheme of traceability. Such a scheme must be based on a material that has similar stability to carbon, but which has just enough reactivity to enable it to link into a chain of reaction schemes with other chemical substances in order to create an unbroken chain of traceability.
The need for such a 'practical mole' is not new; early in the last century in 1912 the analysts working within Nobel's Explosive Company in Britain, one of the founding companies of Imperial Chemical Industries (ICI), put in place a system of reference materials to standardise all the volumetric titrations that formed the core their analytical methods.
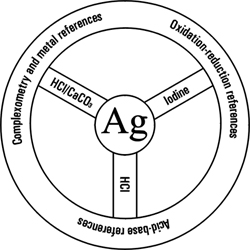 Silver: so many useful reactions
Silver: so many useful reactionsThe system used a sample of specially pure silver as the 'ultimate reference' and had a hierarchy of pure substances as reference materials. Analytical chemists well know that silver can be obtained in a very pure form. It is sufficiently stable over a very long period of time such that its chemical properties do not change. And importantly, it provides the vital link into chemical reactions through its solubility in acids, which can then be reacted with bases and halides. The details of this groundbreaking scheme are recorded in an address given by the President of the Society of Public Analysts on the occasion of their Annual Dinner in London in 1930.
It was this level of attention to analytical excellence, which helped build the strength of ICI over the next 50 years. The concept was taken up other British companies, including the laboratory chemical firm of Hopkin and Williams who used a similar scheme at their Dagenham site near London in the 1970s.
The proper implementation of this type of scheme of traceability involves a great deal of work; calibration analyses must be performed to a higher level of precision than that to which the routine analyses are done. Also, the calibrations should be regularly performed, say once a year, to ensure that routine results remain within set tolerances in the calibrated scheme. Even so, the overwhelming advantage to laboratories that worked with the silver scheme was that their results withstood close scrutiny with others working with similar schemes, no matter where they were located.
 PrimAg: a very practical mole!
PrimAg: a very practical mole!Despite the clear advantages there were many barriers to introducing a silver scheme into many laboratories, not least the availability of resources in terms of time and analytical expertise. As cost saving pressures grew during the late 1980s the system's popularity began to fade and it looked as though another great British development would become part of history.
The early 2000s saw the introduction of the ISO 17025 international standard for the competence of testing and calibration laboratories. Although this was a move in the right direction for chemical metrology, since it required for the first time that CRMs should be traceable to an SI unit, it was nevertheless a challenge for most laboratories.
Which is where ROMIL PrimAg® came in. We wanted to provide a range of metal and ion solution and pure substance CRMs that demonstrated direct traceability to the SI unit, the mole. But we wanted to do so in a way that fitted in with the ROMIL concept of not following established trends and so doing things better, but differently!
 PrimAg: primary reference silver
PrimAg: primary reference silverSo we adapted the well-established concept of silver traceability to the requirements of modern analytical chemistry. Working quietly on our own we proved to ourselves that this was a concept that had been waiting for its time; the adoption of ISO 17025. More importantly we were able to prove to the rigorous technical assessors at the United Kingdom Accreditation Service (UKAS) that our concept and our chemistry were ready for the 21st century. This culminated with our accreditation to ISO 17025 as a chemical calibration laboratory in June 2002. Indeed, we became the first commercial producer worldwide of such CRMs.
Since then quality fashions in the laboratory field have evolved. Today ISO 17034 is the accepted international standard for the competence of reference material producers. Not only does it cover the metrological and traceability requirements of ISO 17025 but also requires assessments of stability, storage and homogeneity of CRMs. Thus, it is not enough that a CRM is characterised with a result at the time of manufacture, but that that result is maintained throughout the period of validity of the CRM, or shelf-life, and within defined storage parameters. Furthermore, the effects of temperature extremes during transport must also be assessed. The results of these assessments must be reflected in the uncertainty estimate associated with the calibration result of the CRM.
In keeping with fashion, ROMIL too have evolved, gaining UKAS accreditation as a reference material producer to ISO Guide 34 in November 2012. Even reference materials that are intrinsically difficult or cannot be traced to silver are covered with the result that all our PrimAg® branded reference materials are produced to this standard. April 2019 saw our transition to the more rigorous ISO 17034 standard.
As for the silver traceability scheme pioneered more than a century ago, it came full circle with our introduction in 2015 of PrimAg®‑TvR traceable volumetric reagents accredited to ISO Guide 34, again a world first, and now accredited under ISO 17034.
As the base for our CRM production, we have retained our original ISO 17025 calibration accreditation, modified to cover our scheme of traceability to ultra-pure silver as a realisation of the SI unit, the mole. Whilst other 17034 reference material producers have base accreditation to ISO 17025 as testing laboratories, we are the only one with the more demanding calibration requirements. We like to think of ourselves as the United Kingdom 'centre of excellence', indeed the global source, for reference materials traced to primary silver.
With such credentials we continue to aspire to be an unbeatable source for the supply of important inorganic solution and solid CRMs to the analytical community.
PrimAg is a registered trademark of ROMIL Ltd.